 |
|



October 27, 2020
This report from the Missouri Hospital Association is designed to help you stay abreast of recent developments related to COVID-19.
Share our online form with individuals interested in receiving this update.
Advocacy
Government Action
Education
Hospital Operations
Noteworthy
Vaccine Updates
Workforce |
|
Dashboard Spotlight
|
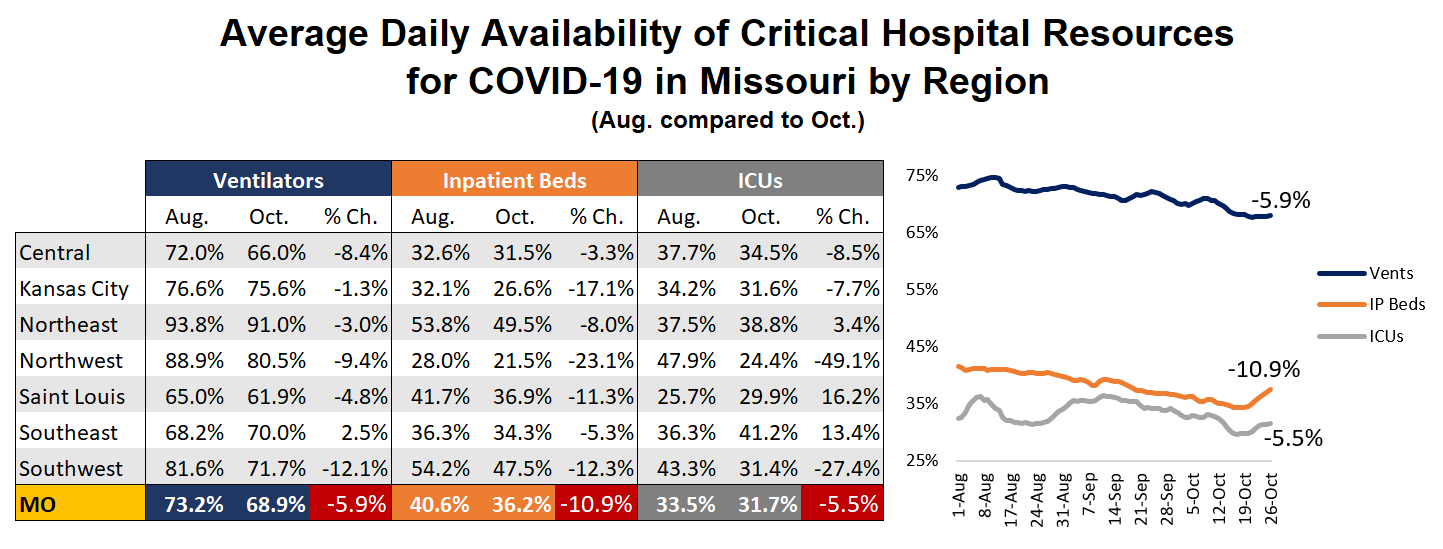
Availability Of Critical Hospital Resources For COVID-19 Patients Steadily Declines
Staff Contact: Jackie Gatz or Mat Reidhead
Since August, the statewide availability of inpatient beds, intensive care units and ventilators has trended downward moderately with significant variation at the regional level. For the state as a whole, the average reported daily availability of mechanical ventilators has decreased by 5.9%, inpatient bed availability has decreased by 10.9% and ICU availability fell by 5.5%. The decreases have been most pronounced in the Southwest and Northwest Regions. |
 |
|
|
Advocacy
|
Congressional Letter To Azar Addresses COVID-19 Testing Costs
 Staff Contact: Daniel Landon or Andrew Wheeler Staff Contact: Daniel Landon or Andrew Wheeler
Congresswoman Ann Wagner of Missouri joined 53 of her U.S. House colleagues in a letter to U.S. Department of Health and Human Services Secretary Alex Azar. It calls for HHS to clarify earlier agency guidance “so that individuals who need a test can receive one without cost-sharing, medical management or prior authorization, and regardless of whether the individual is symptomatic.” The letter notes “in recent weeks, there has been an increase of coverage denials from health plans for COVID-19 testing, particularly for asymptomatic individuals.”
Back To Top
MHA Submits Comments On Interim Final Rule
Staff Contact: Sarah Willson
MHA submitted comments to the Centers for Medicare & Medicaid Services on the interim final rule related to hospital data collection and reporting during the COVID-19 pandemic response. MHA urged CMS to withdraw compliance enforcement actions related to hospital data reporting. The comments reflect member feedback and MHA’s experience working with the agency to understand reporting requirements. Hospitals are encouraged to submit comments before the closing deadline of 4 p.m. CST Monday, Nov. 2.
Back To Top
|
|
Government Action
|
Missouri To Receive Nearly 2 Million Rapid Tests
Staff Contact: Jackie Gatz
The Trump administration is sending 150 million Abbott BinaxNOW COVID-19 tests nationally “to assist governors’ ongoing reopening efforts.” Missouri is receiving 1,840,000 rapid tests, of which 1 million were shipped on Monday. The tests, which are able to detect COVID-19 in as little as 15 minutes, will be distributed to K-12 schools and institutes of higher education, with a portion also retained for state priorities.
Back To Top
HHS Expands List Of Providers Eligible For COVID-19 Relief Funds
Staff Contact: Andrew Wheeler
The U.S. Department of Health and Human Services added residential treatment facilities, chiropractors and vision care providers to the list of providers eligible to receive the latest tranche of COVID-19 Provider Relief Funds. The announcement also references changes to Provider Relief Fund repayment standards, which were previously reported.
Back To Top
Reminder: Relief Funds Available For COVID-19 Testing And Treatment Of Uninsured
Staff Contact: Andrew Wheeler
The Families First Coronavirus Response Act and the CARES Act have set aside funds to help hospitals who test and treat patients for COVID-19. Providers who have conducted COVID-19 testing or provided treatment for uninsured individuals with a COVID-19 primary diagnosis on or after Feb. 4 can request claims reimbursed at Medicare rates. Hospitals receiving reimbursement under the FFCRA or CARES Act uninsured payment cannot balance bill the patients. The Health Resources and Services Administration has a website with details about how to submit claims.
Back To Top
|
|
Education
|
Relias Hosts Webinar On Combatting Burnout In Health Care Providers
Staff Contact: Jill Williams
Relias is hosting a webinar, “Beyond Burnout: Understanding and Combatting Moral Injury in Healthcare Providers,” at 1 p.m. CDT Wednesday, Oct. 28. During the webinar, participants will learn how to identify the signs, symptoms and environmental factors that lead to moral injury for health care workers, compare the differences between how burnout and moral injury manifest and present in health care workers, and apply at least one strategy for how to assess and address potential moral injury.
Back To Top
|
|
Hospital Operations
|
MHA Produces Toolkit For Expanding Hospital Capacity
Staff Contact: Sarah Willson
MHA published a COVID-19: Expanding Hospital Capacity Toolkit to help hospitals plan for and manage surges of COVID-19 patients. As the pandemic response continues, coupled with the resumption of elective surgeries and procedures; increased patient confidence with resuming health care activities; and changes in post-acute testing and tracing activities; the capacity within the hospital may become limited for periods of time. This document focuses on capacity building and patient throughput processes to continue care for an increased volume of patients. While this document focuses on increasing physical capacity, MHA also offers considerations for maximizing workforce in the COVID-19 Surge Staffing Solutions Fact Sheet.
Back To Top
|
|
Noteworthy
|
CDC Publishes Analysis On COVID-19 Hospitalizations Among Health Care Personnel
Staff Contact: Terrie Bauer
The Centers for Disease Control and Prevention published a Morbidity and Mortality Weekly Report on COVID-19-associated hospitalizations among health care personnel. The analysis of COVID-19 hospitalization data from 13 sites indicated that 6% of adults hospitalized with COVID-19 were health care professionals. Among those hospitalized with COVID-19, 36% were in nursing-related occupations and 73% had obesity. Approximately 28% of these patients were admitted to an intensive care unit, 16% required invasive mechanical ventilation and 4% died.
Back To Top
|
|
Vaccine Updates
|
Moderna Fully Enrolls Phase 3 Trial For COVID-19 Vaccine Candidate
Staff Contact: Terrie Bauer
Moderna Inc. announced the completion of enrollment for its COVID-19 vaccine phase 3 trial, confirming 30,000 participants for an efficacy study of mRNA-1273. Moderna and the National Institute of Allergy and Infectious Diseases developed the candidate, which generated a strong immune response in older adults during its phase 1 clinical trial.
Communities that historically have been under-represented in clinical research and are disproportionately impacted by COVID-19 have significant representation among the trial enrollees. Moderna said the study includes more than 11,000 participants from communities of color, representing 37% of the study population and similar to the diversity of the U.S. at large. This includes more than 6,000 participants who identify as Hispanic or Latinx, and more than 3,000 participants who identify as Black or African American.
Back To Top
Pharmacy Technicians, Interns May Administer COVID-19 Vaccines, Tests
Staff Contact: Terrie Bauer
State-licensed pharmacists and state-authorized pharmacy interns under their supervision may administer COVID-19 vaccines and tests authorized or licensed by the U.S. Food and Drug Administration, subject to certain requirements, the U.S. Department of Health and Human Services said in guidance this week. The guidance also clarifies that pharmacy interns authorized under earlier guidance to administer vaccines to children need not be licensed or registered with the state board of pharmacy.
Back To Top
|
|
Workforce
|
Brave Of Heart Fund Available To Families Of Health Care Workers
Staff Contact: Mary Becker
The Brave of Heart Fund was created by the Foundations of New York Life and Cigna with charitable partner E4E Relief. The Brave of Heart Fund provides financial grants to families of eligible health care workers, volunteers and support staff who lost their lives in the fight against COVID-19. For a list of eligible workers, please see the FAQs.
To date, the fund’s activity includes more than $5.4 million in charitable grants that have been awarded to more than 180 families. The grants provide basic and continuing financial support for common needs such as funeral costs, medical care, counseling, food, education, mortgage/rent payments and immediate living expenses. The grantees primarily have been the spouses and dependent children of deceased health care workers and volunteers.
Back To Top
|
 |












 Staff Contact:
Staff Contact: